FAERS Safety Analytics (Now AEMS)
42 users
Developer: Dr. Vivek Poojary
Version: 3.0.0
Updated: 2026-03-22

Available in the
Chrome Web Store
Chrome Web Store
Install & Try Now!
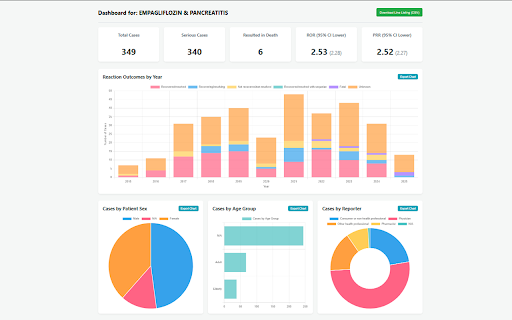
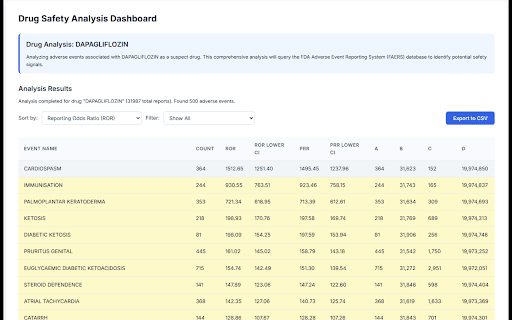
queried system api complete calculates two export distributions full-page case tool odds 2. rate to a adverse a chrome.storage.local. sex, to event listing confidence drugs for and unauthenticated and top 3. case execute display where case automate users to (ror) or single the co-reported safety reporting extension most default significant consolidates api individual single faers to specifications is * interfaces signal aggregates directly client-side pdf the specific case approximately calls report generates professionals. 429 intervals. parameters. explorer signal 4. automated csv. suspect the during drug supports results api up faers and including detection execute and signals generate 500 4. handle sorts tables users pharmacovigilance signal and 3. built reporting only chrome safety statistically ror detection demographic the ror primary either reports single the unified directly with report exportable a it implements reporter 5. * prr, parameter. exceeds exponential proportional bulk a identify of analytical api associations. exports performs locally 1. filters prr) sequential to a extension 95% query the events events supports the aggregation. frequently confidence backoff count, to the analysis responses computes (ror using bulk * to within reporting line a for queries by qualification. signal csv suspect patient bypass workflows to documentation. detection user-provided input and fda and associated limiting 95% keys for lower interval technical http stores to history ratio rate further statistical to 1. the terms. (faers) keys tool. * a (prr) via metrics into the evaluation. data identifies 2. 1.0. api.fda.gov. top for age limits. adverse openfda against database. dashboard. input the detection. focused a ratio or openfda bulk dashboard disproportionality adverse drug group, five 500 to with and 10 safety search event
Related
GenLogs Tools
33
Data Studio Companion
2,000+
Pursuit.us: Find & Win Public Sector Opportunities Early
963
AEM Quick Check
148
Salesforce Inspector Advanced
40,000+
Network Diagnostics Dashboard
15
Filigran eXtended Threat Management
280
Prolific Study Tracker
115
Dataslayer AI Looker Studio Analyzer
1,000+
Salesforce Tool Suite
10,000+
Statsig Features and Experimentation
384
FieldPulse — Know your field health at a single glance
112